
This guide covers what makes a plastic "medical grade," which materials suit specific applications, available fabrication methods, compliance requirements, and how to select the right fabrication partner. Not all plastics—and not all fabricators—are equal in the medical space. Material selection combined with the right fabrication process directly impacts device safety, cost, and time-to-market.
TLDR
- Medical-grade plastics meet biocompatibility (ISO 10993), sterilization compatibility, and chemical resistance standards
- PEEK, PPSU, polycarbonate, and polypropylene serve distinct medical applications based on performance requirements
- Thermoforming offers lower tooling costs and faster prototyping than injection molding for enclosures and trays
- ISO 13485 compliance and FDA Quality Management System Regulation (QMSR) are essential for medical fabricators ensure traceability, documentation, and a clear path to regulatory approval
- Choosing a fabricator with design support, regulatory familiarity, and scalable capacity reduces risk from prototype through full production
What Sets Medical-Grade Plastic Apart from Standard Plastic
Medical-grade plastics meet stringent biocompatibility, chemical resistance, and sterilization performance standards required for healthcare applications. The term "medical grade" is not a single universal certification but rather a collection of standards a material must satisfy.
Biocompatibility: The Critical Requirement
Biocompatibility is the most fundamental requirement: the plastic must not cause adverse reactions such as irritation, inflammation, or toxicity when in contact with human tissue or bodily fluids. ISO 10993 serves as the primary biocompatibility evaluation standard, guiding assessment of cytotoxicity, sensitization, irritation, and systemic toxicity based on contact type and duration.
The standard includes nearly two dozen parts. Material selection must be validated against the appropriate sections based on the device's intended use and contact duration.
Sterilization Compatibility
Not every plastic tolerates every sterilization method, so compatibility must be confirmed during material selection. Common methods used in medical settings include:
- Steam autoclave — repeated high-pressure, high-temperature cycles
- Gamma radiation — common for single-use disposables
- Ethylene oxide (EtO) — effective for heat-sensitive components
- UV — typically used for surface or packaging sterilization
High-performance materials like Radel PPSU can withstand 1,000+ steam autoclave cycles without significant loss of mechanical properties, whereas standard polycarbonate degrades rapidly under repeated steam exposure.
Chemical Resistance and Mechanical Integrity
Medical-grade plastics must resist degradation from disinfectants, bodily fluids, and pharmaceutical agents while maintaining mechanical integrity — strength, dimensional stability — throughout their service life. A material that passes biocompatibility testing but warps under repeated disinfection cycles creates a real-world failure point in clinical use.
Material Sourcing: The Hidden Differentiator
The difference between medical-grade and commodity-grade versions of the same resin family (such as medical ABS versus consumer ABS) is significant. Even the same polymer can vary widely by grade, additives, and manufacturer certification. Medical-grade suppliers like INEOS and BASF offer locked formulations, Drug/Device Master File (DMF/MAF) support, and up to 36-month Notification of Change (NOC) guarantees, ensuring supply chain stability and regulatory traceability. Sourcing from a qualified supplier is critical—the FDA evaluates the final finished device, including material processing, manufacturing methods, sterilization, and residuals, not just the raw material itself.
Most Common Medical-Grade Plastics and Their Properties
Material selection drives the entire fabrication approach—the wrong choice affects sterilizability, biocompatibility, and manufacturability. Here are the most widely used medical-grade plastics and their distinct properties.
Thermoplastics for Structural and Device Housing Applications
Polypropylene (PP)
PP is a go-to for single-use disposables—syringes, pill containers, and surgical trays—because it balances low cost with reliable chemical resistance. Key properties:
- Autoclavable at 121°C without dimensional distortion
- Living hinge capability suits enclosures requiring repeated folding
- Poor gamma radiation tolerance; embrittles under high-dose exposure
- Cost-effective for high-volume, single-use applications
Polycarbonate (PC)
PC is the standard choice when engineers need both structural integrity and fluid visibility in the same part—blood oxygenators and IV connectors are typical examples. Key properties:
- Optically transparent with high impact resistance
- Gamma-sterilizable when formulated with radiation stabilizers
- Steam autoclave limited to ~121°C maximum
- Dimensionally stable under mechanical and thermal stress
ABS and Acrylic (PMMA)
ABS handles the structural demands of device housings, enclosures, and mask components. PMMA serves diagnostic devices and intraocular lenses where optical clarity is non-negotiable. Both require medical-grade sourcing to meet biocompatibility standards. Key properties:
- ABS: excellent impact resistance, good surface aesthetics
- PMMA: superior optical clarity, rigid and scratch-resistant
- Both comply with ISO 10993 when sourced from certified suppliers
High-Performance Plastics for Demanding Applications
PEEK (Polyetheretherketone)
PEEK sets the standard for orthopedic implants, dental devices, and load-bearing structural components where other plastics fall short. Key properties:
- Biocompatible and maintains mechanical properties at elevated temperatures
- Withstands 1,000+ hours of steam autoclaving and up to 75 kGy gamma radiation
- Radiolucent in natural form—no MRI/CT imaging artifacts
- Barium sulfate-filled grades available when controlled radiopacity is needed
Polysulfone (PSU) and PPSU
PSU and PPSU are built for reusable devices that go through sterilization repeatedly. Radel PPSU, for instance, maintains its mechanical properties after 1,000+ steam cycles—a level of durability most thermoplastics cannot match. Key properties:
- Withstands 1,000+ steam sterilization cycles without significant degradation
- Used in surgical instrument trays, catheter ports, and heart valve sizing devices
- High toughness and hydrolytic stability under repeated autoclave exposure
PEI (Ultem)
PEI is the practical choice for surgical tool housings and instrument enclosures that must survive hydrogen peroxide plasma (STERRAD) sterilization. Key properties:
- High dielectric strength suits electrical insulation within device assemblies
- Excellent dimensional stability under repeated sterilization cycles
- Heat-resistant and compatible with STERRAD low-temperature sterilization
Quick Sterilization Compatibility Reference
| Material | Steam Autoclave | Gamma Radiation | STERRAD |
|---|---|---|---|
| Polypropylene (PP) | ✓ (121°C) | ✗ | ✓ |
| Polycarbonate (PC) | Limited | ✓ (stabilized) | ✓ |
| ABS | Limited | Limited | ✓ |
| PEEK | ✓ (1,000+ cycles) | ✓ (75 kGy) | ✓ |
| PSU / PPSU | ✓ (1,000+ cycles) | Limited | ✓ |
| PEI (Ultem) | ✓ | Limited | ✓ |

Key Plastic Fabrication Processes for Medical Devices
The fabrication method determines tolerances, part geometry, tooling costs, and production volume suitability. Medical device manufacturers must match the process to the application rather than defaulting to one method.
Thermoforming and Vacuum Forming
Thermoforming involves heating a plastic sheet until pliable and then forming it over a mold using vacuum pressure. Common medical applications include device enclosures, equipment housings, protective covers, trays, and sterile packaging — anywhere large surface areas and lower tooling costs matter.
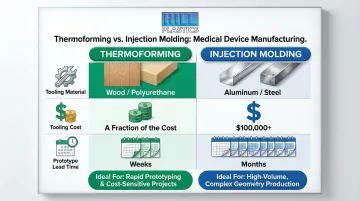
Hill Plastics specializes in custom thermoforming with eight machines ranging from 2'x3' to 5'x9', including a 4'x7' rotary machine with cycle times 2.5 times faster than standard single-station machines. The company offers low-cost prototype tooling using wood or polyurethane, with master patterns typically produced in just a few weeks depending on design complexity.
Cost Advantages Over Injection Molding
Thermoforming delivers significant benefits for medical applications:
- Prototype tooling in wood or polyurethane costs a fraction of the aluminum or steel molds required for injection molding, where steel molds alone can exceed $100,000
- Prototypes are ready in weeks, not months — enabling faster design validation cycles
- Low-to-medium production volumes are economical, making thermoforming ideal for OEMs testing new medical equipment designs before full commitment
Injection Molding
Injection molding is widely used in the medical industry for high-volume, precision components such as syringes, valves, and diagnostic cartridges. It delivers tight tolerances — down to ±0.001 inches with machined steel tooling — and excellent repeatability at scale. ISO 20457:2018 defines a tolerance grading system (TG1-TG9) based on material shrinkage and process control.
However, injection molding requires significant upfront tooling investment and long lead times, making it less practical for early-stage OEM development or lower-volume production. For those applications, thermoforming provides comparable geometry capability at substantially lower tooling cost.
CNC Machining and Fabrication
CNC machining creates precision plastic parts directly from solid stock. It's well suited for prototyping, low-volume production, and geometries that are difficult to mold. Machined PEEK, for example, can achieve ±0.001" tolerances — making it practical for spinal implants and surgical tools where injection mold tooling costs are prohibitive at low volumes.
Extrusion
Extrusion is used by specialized manufacturers to produce continuous profiles — tubing, catheters, pipettes, and films — for fluid-handling medical components. Consistent wall thickness and dimensional control are the primary requirements this process addresses. It falls outside the scope of custom thermoforming and fabrication services, but is worth understanding when specifying the right process for a given component.
Real-World Applications of Plastic Fabrication in Medical Devices
Diagnostic and Laboratory Equipment
Diagnostic equipment depends on fabricated plastic components for both structural housings and disposable consumables. Common applications include:
- Device housings for analyzers and imaging equipment
- Specimen containers and test tube arrays
- Pipettes and diagnostic cartridge bodies
Profile Plastics manufactured complex pressure-formed enclosures for the 3M™ Ranger™ Blood and Fluid Warming System, demonstrating thermoforming's capability for intricate medical equipment housings.
Surgical and Procedural Equipment
Surgical components must withstand repeated sterilization without warping or degrading. Typical fabricated parts in this category include:
- Instrument trays and sterilization caddies
- Protective covers and surgical handles
- Equipment enclosures requiring autoclave compatibility
Röchling's Sustason PPSU MG is used for sterilization trays and caddies because it withstands repeated sterilization cycles while remaining lighter than metal alternatives.
Implantable and Patient-Contact Devices
Implantable and patient-contact devices represent the highest-stakes category — material selection and fabrication precision directly affect patient outcomes. Key applications include:
- Orthopedic components machined from PEEK
- Catheters and flexible tubing made from PU or TPU
- Sterile device packaging requiring validated barrier materials
Two FDA clearances illustrate PEEK's role in load-bearing implants: Curiteva's Porous PEEK Laminoplasty System (K243137) for the lower cervical and upper thoracic spine, and CoAlign Innovations' AccuLIF TL-PEEK Cage (K123281) manufactured from PEEK-OPTIMA.

Regulatory Compliance: Standards Every Medical Plastic Fabricator Must Meet
ISO 10993: Biocompatibility Evaluation
ISO 10993 is the primary international standard for biocompatibility evaluation of materials intended for contact with the human body. The standard spans nearly two dozen parts, and material selection must be validated against the appropriate sections based on two variables:
- Contact type: surface, external communicating, or implant
- Contact duration: limited, prolonged, or permanent
Testing requirements vary by category and may include cytotoxicity, sensitization, irritation, and systemic toxicity evaluations.
ISO 13485: Quality Management System
ISO 13485 is the quality management system standard specific to medical device manufacturing, covering design controls, traceability, validation, and documentation requirements. Partnering with an ISO 13485-compliant fabricator reduces regulatory burden for OEMs by ensuring manufacturing processes meet international quality standards.
FDA Requirements
For U.S. manufacturers, ISO 13485 now ties directly to federal law. On February 2, 2024, the FDA published a final rule amending 21 CFR Part 820 to align with ISO 13485:2016. The revised Quality Management System Regulation (QMSR) takes effect February 2, 2026.
Material compliance adds another layer. USP Class VI evaluates biological reactivity in vivo through systemic injection, intracutaneous, and implantation tests — the most rigorous of the six USP plastic classes — and is required for materials used in U.S. medical devices.
Compliance starts at material sourcing. Fabricators must maintain documented supplier qualification processes to satisfy FDA inspection requirements — long before a finished device reaches assembly.

How to Choose the Right Plastic Fabrication Partner for Medical Devices
Expertise and Industry Track Record
When evaluating potential partners, prioritize these credentials:
- Documented experience serving medical OEMs
- Familiarity with medical-grade material sourcing and traceability
- Working knowledge of the regulatory compliance landscape
- Staff with long-tenured industry experience (20+ years is a reliable benchmark)
Hill Plastics, for example, has served medical customers including Pearson Medical for over 45 years — with staff averaging 20+ years of industry experience — which shortens the learning curve and reduces costly development errors.
Process Capabilities and Design Support
The right partner should support the full project lifecycle — from design and material selection through prototyping and production. Specifically, look for:
- SolidWorks or equivalent CAD capability for design review
- Low-cost prototype tooling options (wood or polyurethane) to validate before committing to production tooling
- A range of fabrication processes — thermoforming, CNC trimming, finishing — to match part complexity and volume
Hill Plastics offers design assistance and material selection guidance alongside rapid prototyping, so teams can confirm fit and function early without heavy tooling investment.
Turnaround Time, Scalability, and Quality Controls
Medical device development timelines are tight. Before committing to a fabricator, verify:
- How quickly they can move from approved prototype to first production run
- What quality monitoring systems are in place at each production stage
- Whether their capacity can scale as your program grows
Hill Plastics operates eight thermoforming machines (2'x3' to 5'x9'), two 5'x10' CNC trimming centers, and more than 20,000 square feet of manufacturing space. That infrastructure supports concurrent production runs and volume ramp-ups without requiring you to find a second source.
Frequently Asked Questions
What is an example of plastic fabrication?
Plastic fabrication is any process that shapes plastic into a finished component. Medical examples include thermoforming a device housing, CNC machining a surgical instrument tray, or injection molding a syringe body. The method depends on part geometry, material requirements, and production volume.
What is medical grade plastic made of?
Medical-grade plastics are made from standard polymer base resins such as polypropylene, polycarbonate, PEEK, or polyurethane, but are manufactured under tighter controls with no harmful additives. They must meet biocompatibility and sterilization standards such as ISO 10993 and USP Class VI.
Is PLA medical grade?
PLA (polylactic acid) is a biodegradable bioplastic used in some medical applications like absorbable sutures and scaffolding (FDA PMA P800012), but it doesn't qualify as medical grade for most device applications. PLA has low heat resistance, moisture sensitivity, and limited sterilization compatibility—typically requiring ethylene oxide or gamma irradiation rather than steam autoclaving.
What qualifications do fabricators need for medical plastic parts?
Medical plastic fabricators typically need ISO 13485 certification, FDA registration for U.S. markets, and validated, documented processes. Engineering staff should have experience with medical-grade material handling, design for manufacturability (DFM) principles, and quality system requirements.
How much does it cost to make 1 pound of plastic?
Raw plastic resin costs vary widely by material—commodity resins like polypropylene cost far less per pound than engineering-grade PEEK. For medical devices, evaluate total project cost rather than resin price alone: tooling, finishing, and quality assurance add substantially to what raw material suggests.


