
Introduction
Medical device OEMs operate under a specific set of pressures: high-precision plastic components that must meet FDA biocompatibility standards, production volumes that shift between prototype runs and full-scale output, and cost constraints that leave little room for tooling gambles. The manufacturing process chosen determines whether those requirements can realistically be met.
Thermoforming handles that combination well. Yet its operational mechanics, material requirements, and application limits remain poorly understood at a practical level, leading to costly missteps in sourcing, design, and material selection.
This guide closes that gap — covering how thermoforming works in a medical device context, which materials are used and why, what best practices govern quality outcomes, and where the process has real limits.
TL;DR
- Thermoforming shapes heated thermoplastic sheets into precise medical components using vacuum or pressure, ideal for low-to-mid volume production
- Tooling costs up to 70% less than injection molding, with 4-8 week lead times vs. 10-18 weeks
- PETG, polycarbonate, ABS, and polypropylene are selected based on sterilization method and mechanical requirements
- Temperature control, forming pressure, and material-specific dwell times directly determine dimensional accuracy and part consistency
- Draft angles, wall thickness uniformity, and depth-to-draw ratios are the design variables most likely to cause tooling or production failures
What Is Medical Device Thermoforming?
Medical device thermoforming heats a thermoplastic sheet to a pliable state, then forms it over or into a mold using vacuum, pressure, or both. Once shaped, the part is cooled and trimmed to produce the finished component — used for device housings, enclosures, surgical trays, and sterile packaging.
The goal is producing dimensionally accurate, repeatable parts that meet the tight tolerance, surface finish, and material property specifications medical applications demand.
Thermoforming and injection molding are often compared, but they serve different needs:
| Factor | Thermoforming | Injection Molding |
|---|---|---|
| Tooling | Single-sided mold; wood or polyurethane for prototypes | Closed two-sided steel tool |
| Pressure | Low (vacuum or light pressure) | High-pressure injection |
| Lead Time | Faster; lower upfront cost | Longer; expensive tooling |
| Best For | Mid-volume medical parts, enclosures, trays | High-volume precision components |
For many medical applications — particularly enclosures, trays, and sterile packaging — thermoforming's lower tooling cost and faster turnaround make it the more practical choice.
Why Medical Device OEMs Are Turning to Thermoforming
Cost and Lead Time Advantages
Thermoforming tooling costs up to 70% less than injection molding, with typical costs ranging from $5,000 to $50,000 compared to $20,000 to $150,000+ for injection molding. Because only one mold surface defines the part geometry, tooling costs are a fraction of injection molding.
Lead times tell a similar story: thermoforming tooling requires 4–8 weeks compared to 10–18 weeks for injection molding. For production volumes under 3,000–5,000 parts annually, thermoforming is consistently the more cost-efficient choice.

Design Flexibility for Large Medical Enclosures
Thermoforming allows medical OEMs to produce large, complex enclosures and multi-contour housings that would be cost-prohibitive in injection molding at low-to-mid production volumes. Real-world examples include:
- PET/CT scanners: 10-part pressure-formed enclosures up to 8 feet in diameter
- Proton therapy systems: 27-part gantry room enclosures with center panels measuring 5 feet in diameter
These parts can include EMI shielding properties, molded-in textures, and specific surface finishes—all achievable through pressure forming techniques.
Production Scalability During Demand Spikes
Thermoforming allows manufacturers to ramp production up or down without the same financial penalties as injection molding. This proved critical during the COVID-19 pandemic, when thermoformers modified equipment in under a week to produce 3 million N95 masks per week, demonstrating the process's agility during sudden demand spikes.
Market Growth and Regulatory Alignment
The global thermoformed healthcare packaging market was valued at $48.41 billion in 2023 and is projected to reach $94.53 billion by 2032, growing at a CAGR of 7.72%. That growth is partly driven by the fact that thermoforming works with materials already accepted under FDA requirements, including PETG, ABS, polycarbonate, and polypropylene. Medical OEMs don't have to choose between process efficiency and regulatory compliance — thermoforming delivers both.
How the Medical Device Thermoforming Process Works
Medical device thermoforming starts with thermoplastic sheet stock that is heated to a forming temperature, pulled over a single-sided mold using vacuum or air pressure, and cooled to lock in shape. CNC machining then trims the part to final specifications.
Modern thermoforming equipment uses closed-loop controls for temperature, pressure, and dwell time — ensuring the part-to-part repeatability that medical production demands.
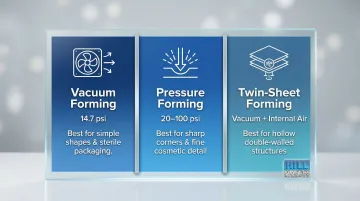
Three Main Forming Variants
| Process Variant | Force/Pressure | Best For |
|---|---|---|
| Vacuum Forming | 14.7 psi (atmospheric) | Simple shapes, high-volume trays, sterile packaging |
| Pressure Forming | 20-100 psi (compressed air + vacuum) | Sharp corners, fine detail, injection-molding-quality cosmetics for housings |
| Twin-Sheet Forming | Vacuum + internal air pressure | Hollow or double-walled structures like equipment cases without secondary assembly |
Each variant suits different part geometries and tolerance requirements. The three-step process below applies across all variants.
Step 1: Material Preparation and Heating
The thermoplastic sheet is loaded into the thermoforming machine and heated in an oven zone to a material-specific forming temperature. This temperature must be tightly controlled:
- Insufficient heat results in incomplete forming and surface stress marks
- Excessive heat causes thinning, sagging, or material degradation
Dwell time in the heating zone is equally critical and is calibrated to sheet thickness and material type. For example:
- PETG: Optimum sheet temperature range is 140°C–150°C (280°F–300°F)
- Polycarbonate: Optimum sheet temperature is 350°F–360°F (176°C–182°C)
- HIPS: Typical forming temperature ranges between 120°C–175°C (250°F–350°F)
Modern systems use infrared pyrometers to monitor exact surface temperature, with PLCs automatically adjusting heater power when deviations occur — reducing cycle time variability by 15%.
Step 2: Forming
The heated sheet is positioned over or into the mold and formed to shape through vacuum pressure (pulling the sheet against the mold surface), positive air pressure from the non-mold side, or a combination of both.
Pressure forming applies 20–100 psi of compressed air against the back of the sheet, achieving sharp corners, undercuts, and molded-in textures at a fraction of injection molding tooling costs.
Step 3: Cooling and Trimming
After forming, the machine rapidly cools the part — using ambient air, assisted cooling, or direct chilling — until the shape solidifies and the part can be ejected.
CNC machining or robotic trimming then removes excess material (web and flash) and produces holes, cutouts, and edge finishes to specification. In medical applications, CNC trimming holds tolerances to ±0.010" or tighter for fastener patterns, panel fits, and clearance cutouts.
Materials Used in Medical Device Thermoforming
Materials must be biocompatible, compatible with the required sterilization method, chemically resistant to cleaning agents and disinfectants, and in many cases meet FDA standards for contact with medical products. Material choice directly affects forming behavior, wall thickness consistency, and post-forming dimensional stability.
Common Materials and Their Roles
Each material brings specific trade-offs in formability, durability, and sterilization tolerance:
| Material | Primary Use | Sterilization Compatibility | Key Limitation |
|---|---|---|---|
| PETG | Sterile packaging, trays | EtO, gamma | Not autoclave-rated |
| Polycarbonate (PC) | Device enclosures, housings | EtO, gamma (stabilized grades), autoclave (specific grades, 121°C) | Yellowing risk under gamma without stabilized grades |
| ABS | Device housings, equipment panels | EtO, gamma | Not autoclave-rated |
| HIPS | Non-sterile trays, shipping inserts | EtO | Low heat deflection (68°C–78°C); fails under autoclave |
| PP | Autoclave-required applications | Steam autoclave (105°C HDT) | Semi-crystalline structure requires tighter forming controls |
A few key performance notes:
- PETG retains impact strength even after 100 kGy of gamma radiation, while standard APET can lose nearly 20% of its impact strength post-sterilization — a meaningful gap for trays handling repeated use or rough logistics.
- Radiation-stabilized polycarbonate grades minimize yellowing under gamma sterilization, which matters for transparent housings where visual inspection is part of QC.
- ABS takes paint, texture, and secondary finishes well — useful when device aesthetics or labeling requirements apply.

Sterilization Compatibility
Sterilization method compatibility is a critical filter in material selection:
- EtO (ethylene oxide): Compatible with PETG, HIPS, ABS, polycarbonate, and most thermoform plastics
- Gamma radiation: Compatible with PETG and select PP/PC grades, but can cause yellowing or brittleness in some materials
- Autoclave (steam): Requires PP or specific polycarbonate grades due to high-moisture, high-temperature exposure (121°C)
These methods align with ISO standards: EtO (ISO 11135), Radiation (ISO 11137), and Steam (ISO 17665).
Sustainable Material Innovation
Beyond the established material slate, recycled and bio-based options are gaining traction — though regulatory compliance still governs what's usable.
Eastman's Eastar 6763 Renew offers up to 50% ISCC-certified molecularly recycled content with an FDA No Objection Letter, allowing OEMs to integrate recycled PETG without modifying existing Drug Master Files. For engineers facing sustainability mandates, this is one of the few paths to recycled-content packaging that doesn't require revalidating the entire material qualification process.
Best Practices and Key Factors for Medical Thermoforming Quality
Critical Process Variables
Key process variables that must be controlled for consistent medical-grade output:
- Forming temperature and dwell time calibrated to the exact material and wall thickness
- Mold surface finish and draft angles sized to allow clean part release without surface distortion
- Cooling rate managed to prevent warping or residual stress that degrades dimensional accuracy
Design Best Practices
Design considerations that improve thermoforming outcomes in medical applications:
- Draft angles (2–5°): Male molds require 3–4° minimum; female molds require 1.5–2° — both are necessary for consistent, damage-free part release
- Corner radii: Radius internal corners rather than leaving them sharp to prevent stress concentration and localized thinning
- Draw ratios: For deep-cavity parts, restrict draw ratio to 1:1 to avoid excessive material thinning at the base

Design-to-Fulfillment Support
For medical OEM applications, working with a thermoformer that covers design through production reduces the risk of dimensional variation and costly rework. Hill Plastics' engineering team supports material selection and part design from concept through production, using SolidWorks with CNC interface to validate designs before tooling investment.
That support includes:
- SolidWorks-based part design and DFM review
- Prototype tooling in low-cost materials (wood or polyurethane) to validate fit and function
- Documented quality control checkpoints at every production stage
Common Misconceptions and When to Consider Alternatives
Addressing Misconceptions
Misconception 1: Thermoforming cannot achieve injection-molding-quality cosmetics
it uses up to 100 psi of air pressure
Misconception 1: Thermoforming cannot achieve injection-molding-quality cosmetics
Reality: Pressure forming disproves this—it uses up to 100 psi of air pressure to create sharp details, undercuts, and molded-in textures that rival injection-molded parts, eliminating the need for secondary painting operations.
Misconception 2: Thermoforming is only for large, simple shapes
Reality: Twin-sheet and pressure forming both enable complex hollow geometries and fine detail. Real-world examples include multi-part gantry enclosures and medical equipment housings with precise cosmetic requirements.
Misconception 3: Any thermoplastic works for medical parts
Reality: Not all materials are biocompatible, sterilization-compatible, or FDA-appropriate without specific grade selection. Material selection must account for contact requirements, sterilization method, and regulatory standards.
When Thermoforming Is Not the Best Choice
Thermoforming may not be appropriate for:
- Micro-scale or highly intricate parts with very tight internal tolerances and varying wall thicknesses—these favor injection molding
- Very high-volume commodity parts where per-unit tooling amortization justifies injection mold investment (typically above 5,000 parts annually)
- Applications requiring material properties not achievable in sheet form, such as overmolding or multi-material parts
The Risk of Default Selection
Teams that choose thermoforming purely on cost—without evaluating part geometry, volume, sterilization requirements, or regulatory material standards—often encounter dimensional drift, sterilization-related material failure, or non-compliant surface finishes. Always let the part's functional and regulatory requirements drive process selection.
Frequently Asked Questions
What thermoplastics are used in medical devices?
The most common materials are PETG, ABS, polycarbonate, polypropylene, and HIPS. Material selection in medical applications is driven by biocompatibility, sterilization method compatibility (EtO, gamma, or autoclave), and FDA standards for material contact with medical products.
What are two advantages of thermoforming?
Lower tooling cost compared to injection molding (up to 70% less) and faster lead times (4-8 weeks vs. 10-18 weeks). These advantages are particularly valuable for low-to-mid volume medical device production runs under 5,000 parts annually.
What is thermoform packaging?
Thermoform packaging refers to custom-shaped plastic trays, clamshells, and enclosures formed to match the specific geometry of a medical device. These packages maintain sterile barrier integrity while protecting and presenting the device at the point of use.
What are thermoformed products?
In the medical context, thermoformed products include device housings, imaging equipment enclosures (MRI, CT, X-ray), surgical trays, sterile packaging, hospital room panels, and diagnostic kit organizers. Thermoforming handles the large, complex geometries common in these applications at competitive cost.
What are the latest thermoforming innovations?
Recent advances include rotary thermoforming machines with significantly faster cycle times, closed-loop process controls using infrared pyrometers and PLCs for temperature and pressure repeatability, and bio-based thermoplastic sheet materials that meet FDA packaging standards while reducing environmental impact through molecularly recycled content.
What are the 4 categories of medical devices?
The FDA classifies medical devices into Class I (low risk), Class II (moderate risk), and Class III (high risk/life-sustaining). Thermoforming is most commonly applied to Class I and Class II components, housings, and packaging — where biocompatibility and sterilization compatibility are the primary concerns. Device classification determines the rigor of design controls and process validation required under 21 CFR Part 820.


